Identify the acid and base which form sodium hydrogen carbonate. Write chemical equation in support of your answer. State whether this compound is acidic, basic or neutral. Also write its pH value.

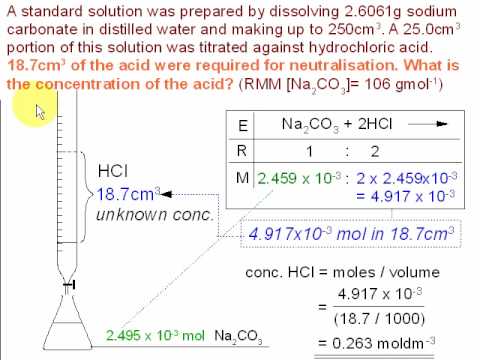
Determination of sodium carbonate and sodium bicarbonate in a solution containing both salts | BPC2113 - General Chemistry - IMU | Thinkswap

SOLVED: DATA AND CALCULATIONS Fill in the table Show your work in the space below the table Sodium Bicarbonate Solution Sodium Carbonate Solution Mass of Solute 0 840 I.Ol 4 12 .

SOLVED: Calculate the pH of an aqueous solution that contains 0.392 M sodium carbonate and 0.177 M sodium hydrogen carbonate. pH =

SOLVED: 0.1Mwith the initial pH of O.O1M 50 mL Na2cO3 sodium carbonate solution Calculate the pH values of the Ist and 2nd turning points during the titration with HCI Ka1 1,8.1016 Ka2

During the titration of sodium carbonate with H Cl, the dissolved carbonate ion will exist in three different forms; CO_3^{-2}, H CO_3^{-1}, and H_2 CO_3. During which part of the titration (initial,






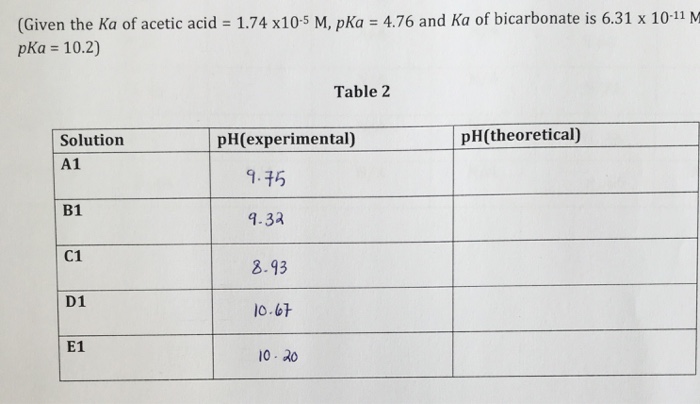
![PDF] A sodium carbonate-bicarbonate buffer for alkaline phosphatases. | Semantic Scholar PDF] A sodium carbonate-bicarbonate buffer for alkaline phosphatases. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/aaa98c1f9de4c398b6a057bf4936ea76736034c7/1-Table1-1.png)