
Calculating p H not that hard - Calculating pH To calculate the pH of an aqueous solution you need - Studocu
![SOLVED: Helpful Equations pH = -log [HzO+ ] Kw = Ka X Kb [ATJ[Hao+1 [HA] = pOH -log [OH:] 14 = pH + pOH [HA][OH-] [A-] Kb Henderson- Hasselbalch Equation [A-] pH =pKa log - [HA] SOLVED: Helpful Equations pH = -log [HzO+ ] Kw = Ka X Kb [ATJ[Hao+1 [HA] = pOH -log [OH:] 14 = pH + pOH [HA][OH-] [A-] Kb Henderson- Hasselbalch Equation [A-] pH =pKa log - [HA]](https://cdn.numerade.com/ask_images/9f4de4f19628424f8aea53cff3ecadcb.jpg)
SOLVED: Helpful Equations pH = -log [HzO+ ] Kw = Ka X Kb [ATJ[Hao+1 [HA] = pOH -log [OH:] 14 = pH + pOH [HA][OH-] [A-] Kb Henderson- Hasselbalch Equation [A-] pH =pKa log - [HA]







![Solved 2. Calculate [H+] and [OH-] for the following: [H* | | Chegg.com Solved 2. Calculate [H+] and [OH-] for the following: [H* | | Chegg.com](https://media.cheggcdn.com/media/b15/b1559073-2f2f-46fe-a023-ac5777fa7482/phpLXeISS)


![Calculating [OH-], pH and pOH from Kb Calculating [OH-], pH and pOH from Kb](https://www.mi.mun.ca/users/pfisher/chemistry1011_135/img007.gif)
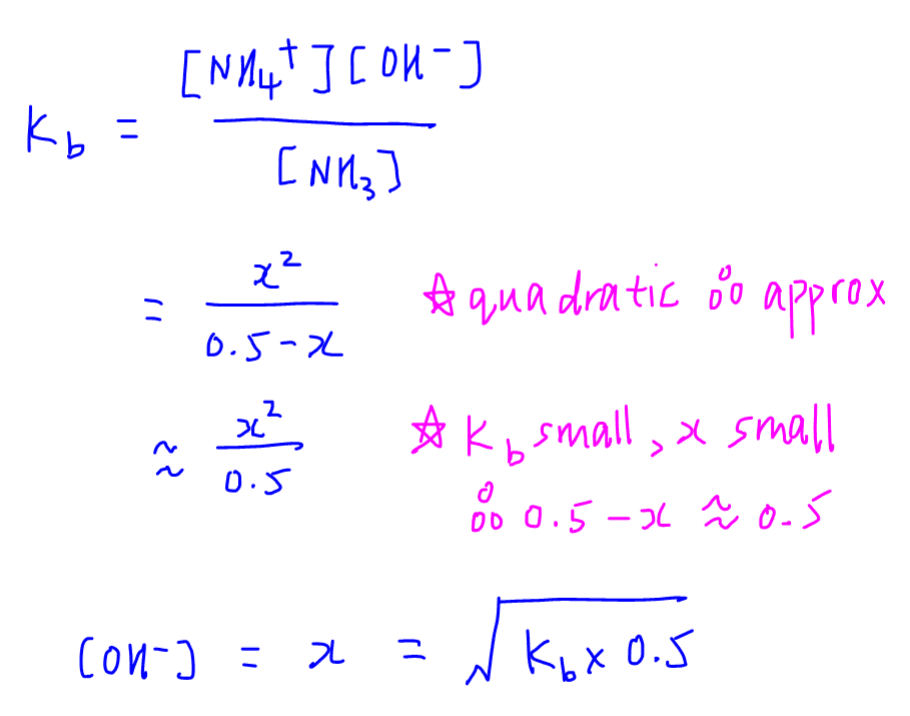






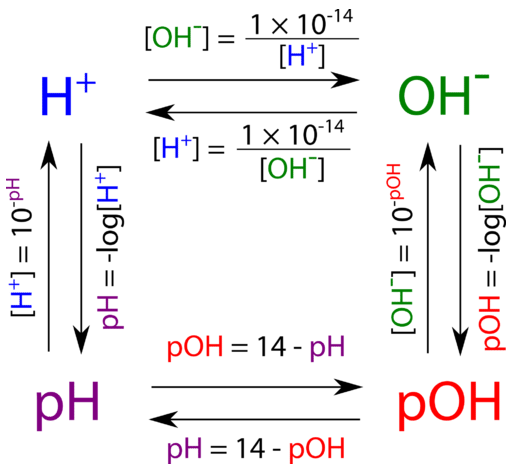
![Calculations of pH, pOH, [H+] and [OH-] Calculations of pH, pOH, [H+] and [OH-]](https://www.sciencegeek.net/Chemistry/taters/graphics/pHSchematic.gif)
![Calculating [H₃O⁺] and pH (worked examples) (video) | Khan Academy Calculating [H₃O⁺] and pH (worked examples) (video) | Khan Academy](https://cdn.kastatic.org/ka_thumbnails_cache/e67920d2-30a0-40ef-bcfb-a7761c9673f6_1280_720_base.png)

![Calculating pH & pOH, [H+], [OH-], Acids & Bases CLEAR & SIMPLE - YouTube Calculating pH & pOH, [H+], [OH-], Acids & Bases CLEAR & SIMPLE - YouTube](https://i.ytimg.com/vi/eoM1nzYFy8k/mqdefault.jpg)
![Calculating pH, pOH, [H+], [H3O+], [OH-] of Acids and Bases - Practice - YouTube Calculating pH, pOH, [H+], [H3O+], [OH-] of Acids and Bases - Practice - YouTube](https://i.ytimg.com/vi/UiK37I159fc/maxresdefault.jpg)
![Calculating pH & pOH, [H+], [OH-], Acids & Bases CLEAR & SIMPLE - YouTube Calculating pH & pOH, [H+], [OH-], Acids & Bases CLEAR & SIMPLE - YouTube](https://i.ytimg.com/vi/eoM1nzYFy8k/maxresdefault.jpg)