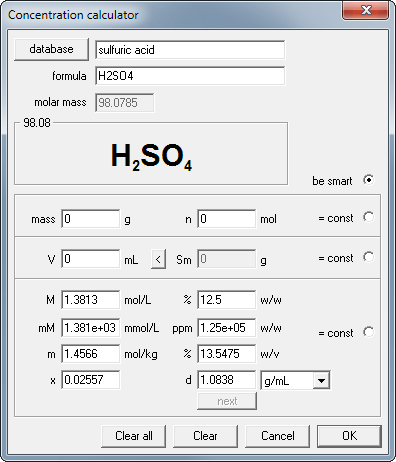
Concentration conversions using CASC - converting molarity to percent concentration, checking density
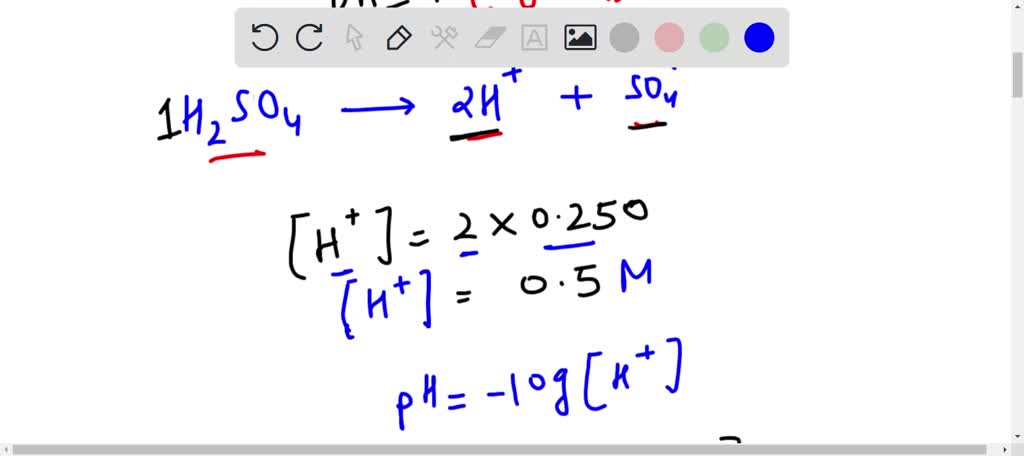
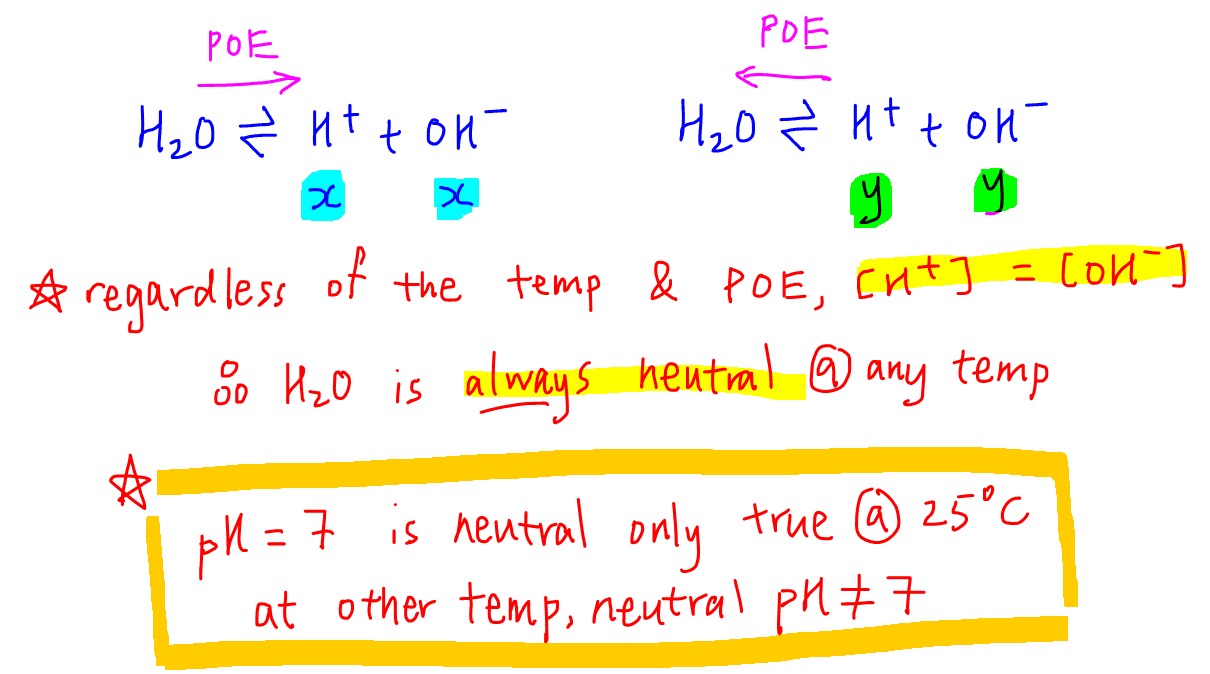
What is the pH value of a solution with 10M of sulphuric acid? If we calculate it, a negative value will be obtained, but pH value cannot be a positive value. -

Curve for pH as a function of sulfuric acid concentration based on the... | Download Scientific Diagram

Calculate the pH of the resultant mixtures:a) 10 mL of 0.2M Ca (OH)2 + 25 mL of 0.1M HCl b) 10 mL of 0.01M H2SO4 + 10 mL of 0.01M Ca (OH)2