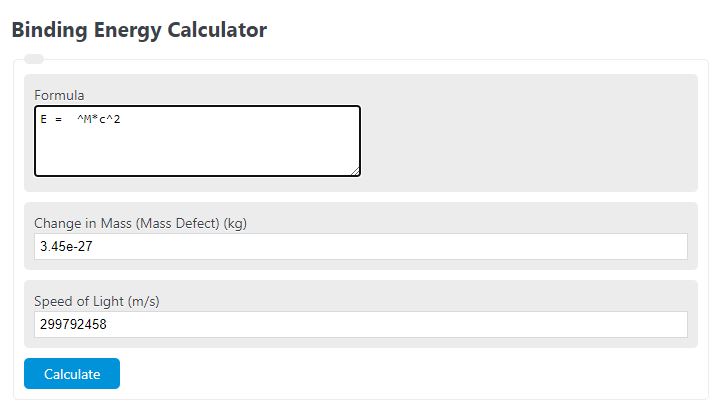
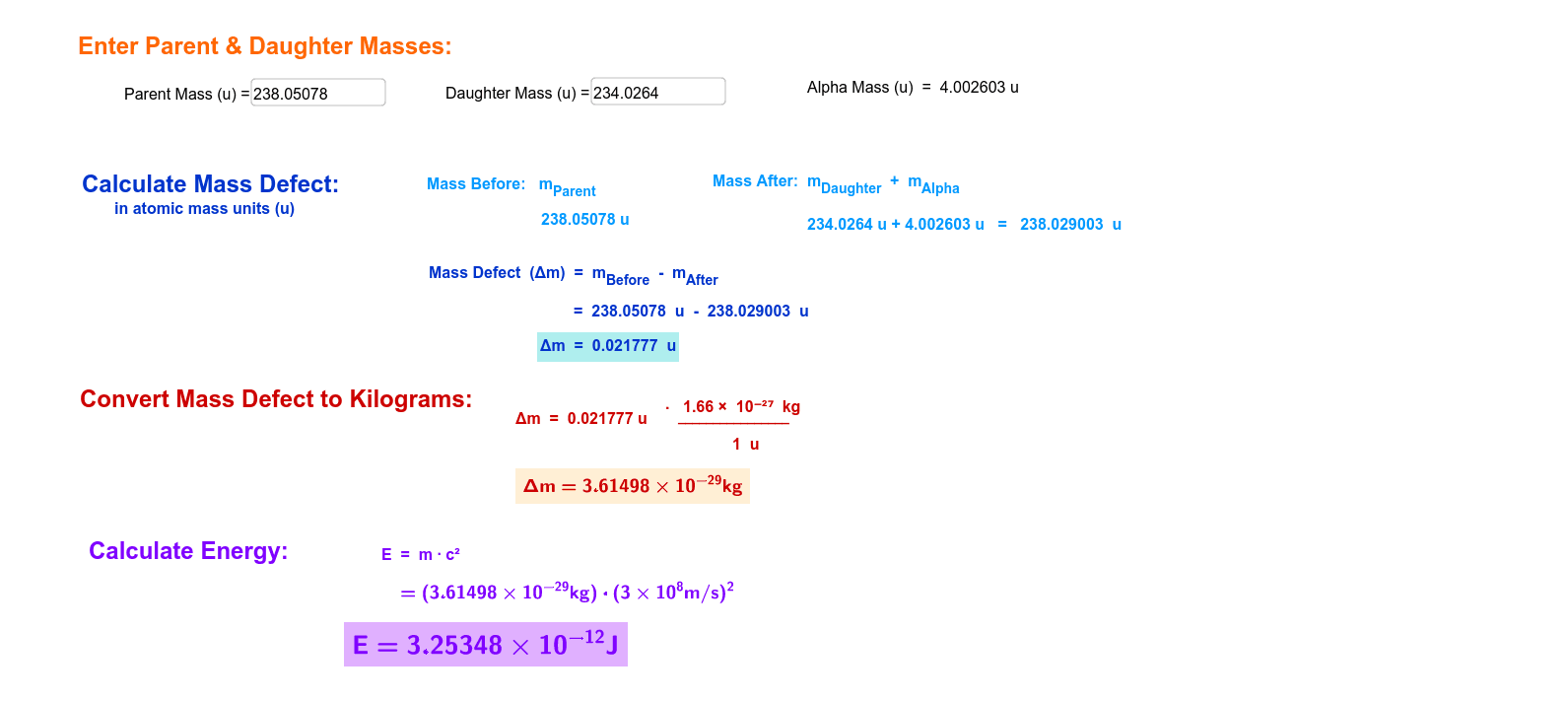
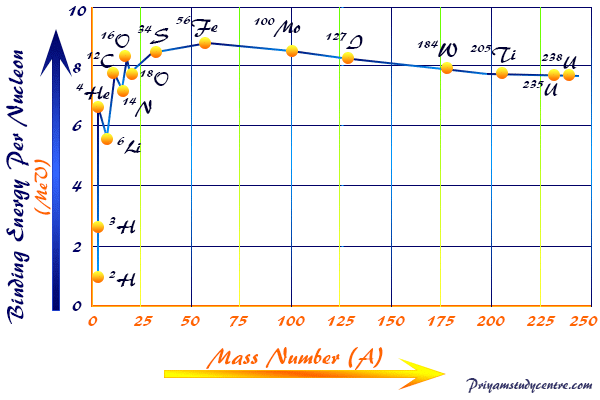
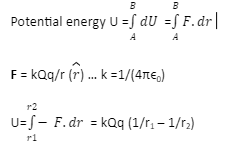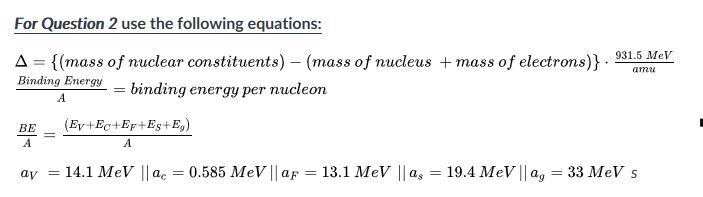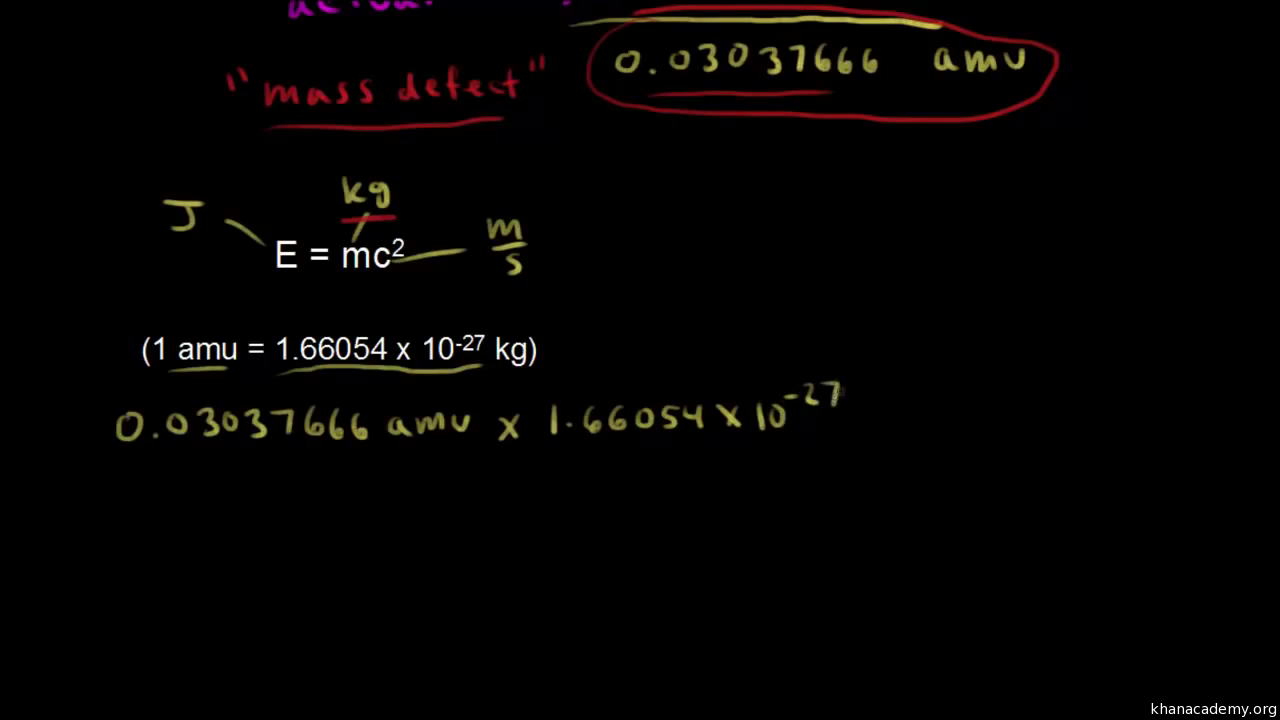Calculate the binding energy and binding energy per nucleon (in MeV) of a nitrogen nucleus (^147N) from the following data :Mass of proton = 1.00783 uMass of neutron = 1.00867 uMass of nitrogen nucleus = 14.00307 u
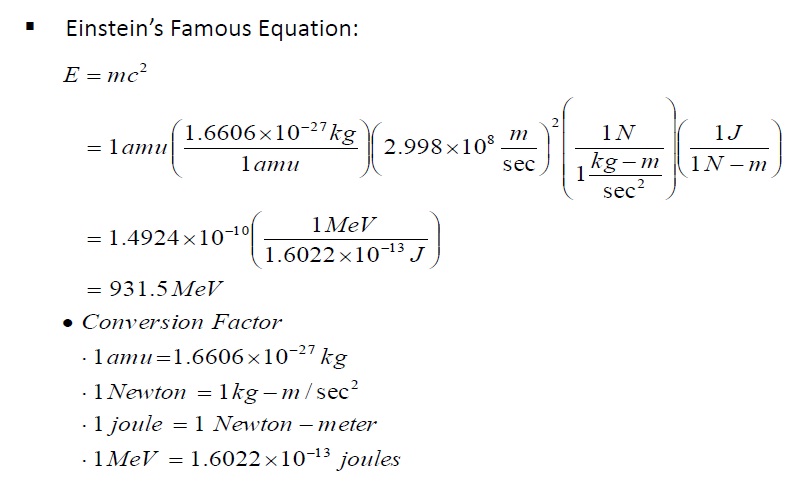
In nuclear physics, what is meant by the term 'mass defect', and what is its relation to nuclear energy? | Socratic

Mass Defect Formula & Examples | What is Nuclear Mass Defect? - Video & Lesson Transcript | Study.com

Calculate the (i) mass defect, (ii) binding energy and (iii) the binding energy per nucleon for a 6C^12 nucleus. Nuclear mass of 6C^12 = 12.000000 a.m.u., mass of hydrogen nucleus = 1.007825

Mass Defect Formula & Examples | What is Nuclear Mass Defect? - Video & Lesson Transcript | Study.com