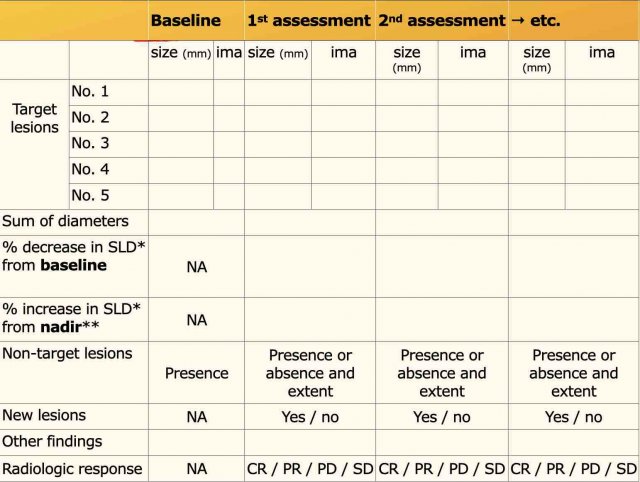
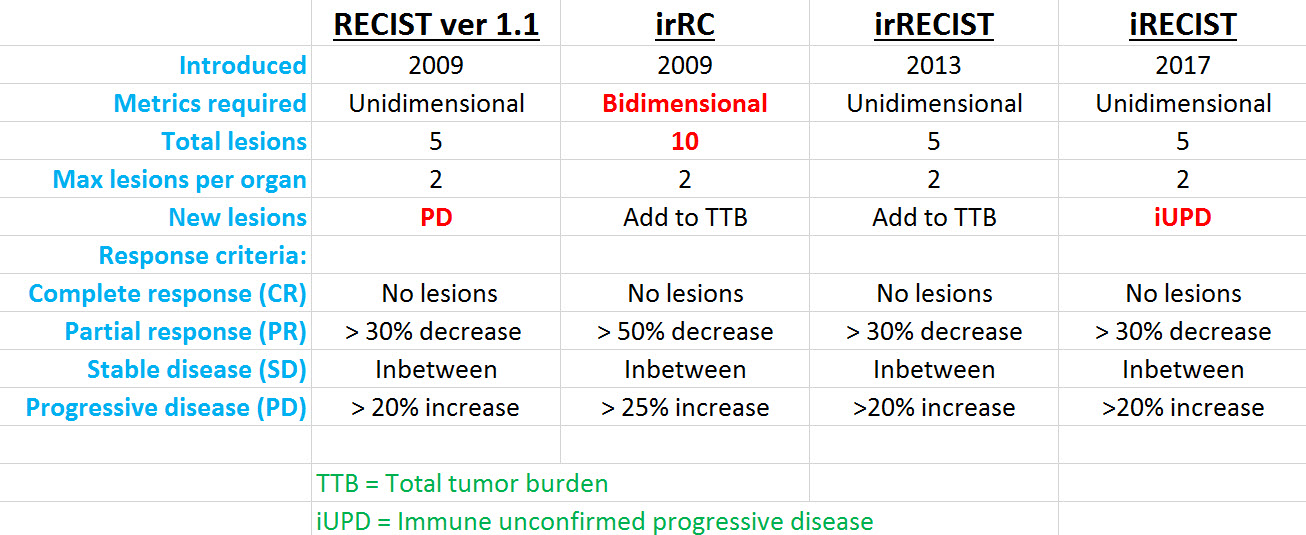
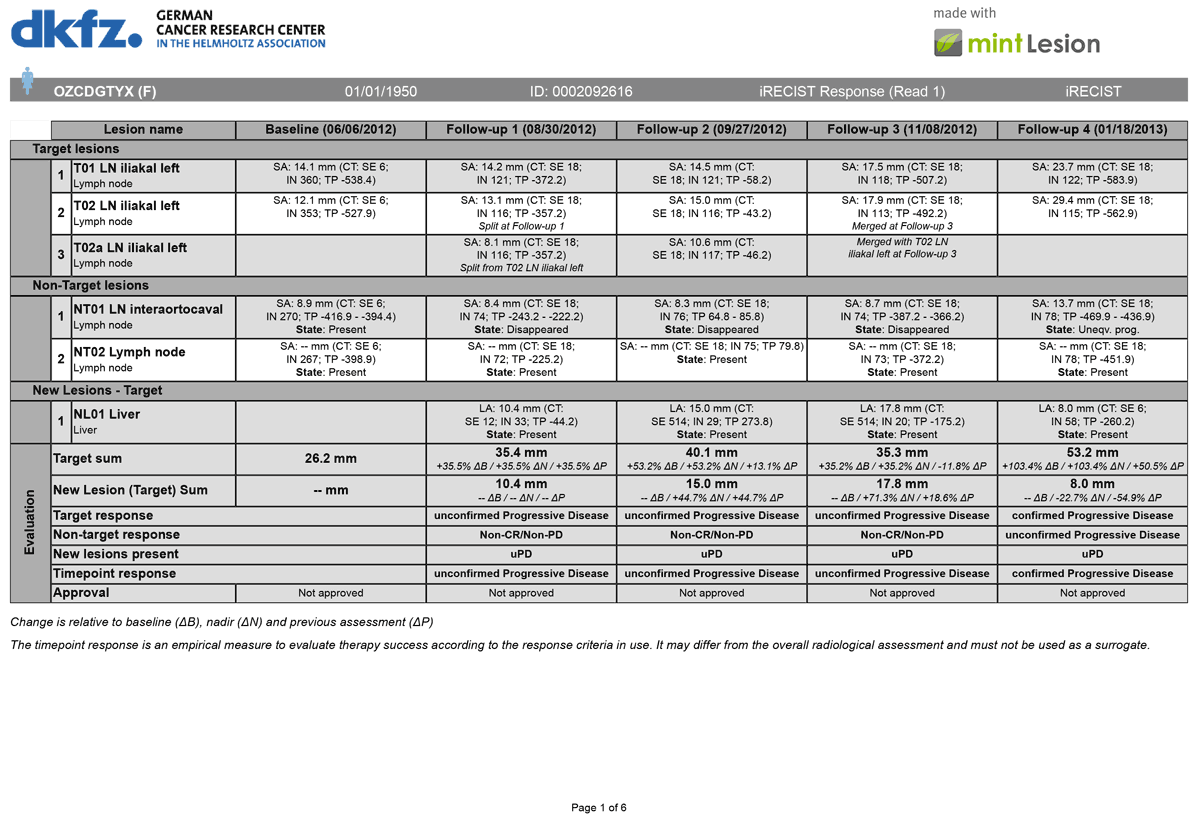
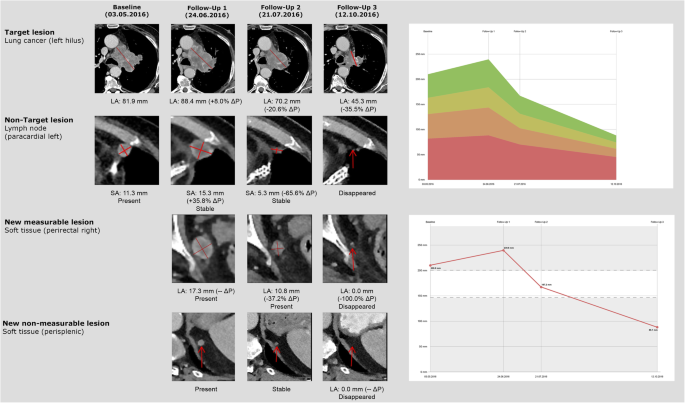
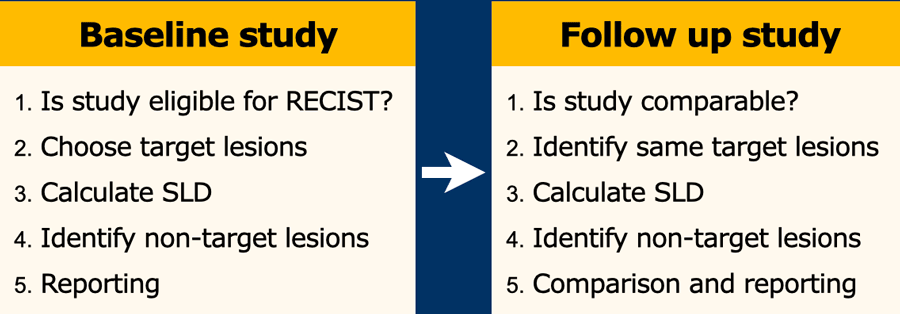
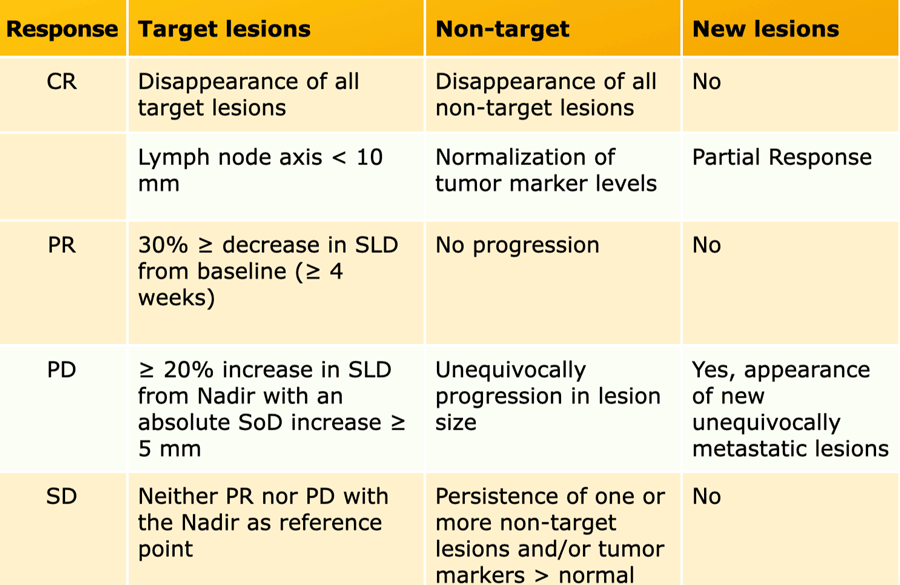
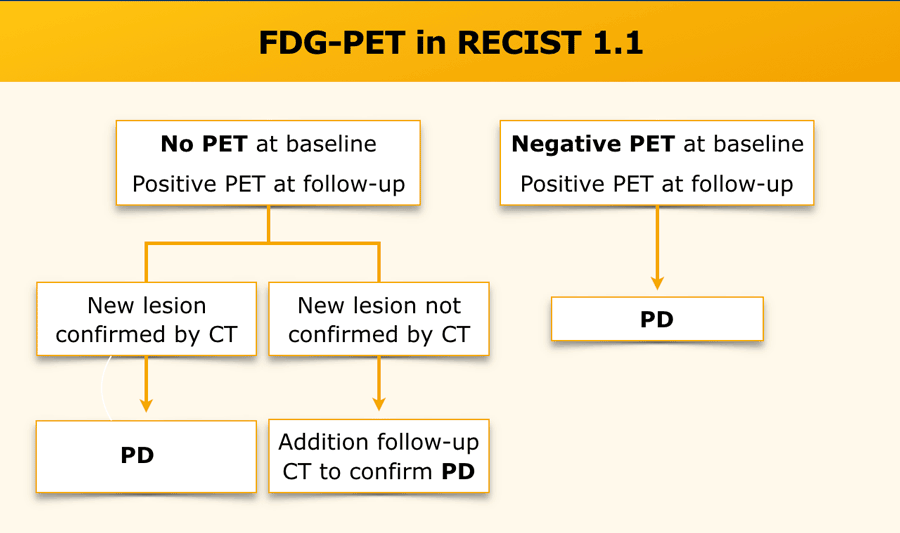
iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics - The Lancet Oncology

iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics - ScienceDirect

PDF) Comparison of iRECIST versus RECIST V.1.1 in patients treated with an anti-PD-1 or PD-L1 antibody: pooled FDA analysis
Statistical Analysis Plan Title: A Phase 1/2, Multicenter, Single-Arm, Open-Label, Dose-Escalation Study of Birinapant in Combin
Simplifying the Derivation of Best Overall Response per RECIST 1.1 and iRECIST in Solid Tumor Clinical Studies
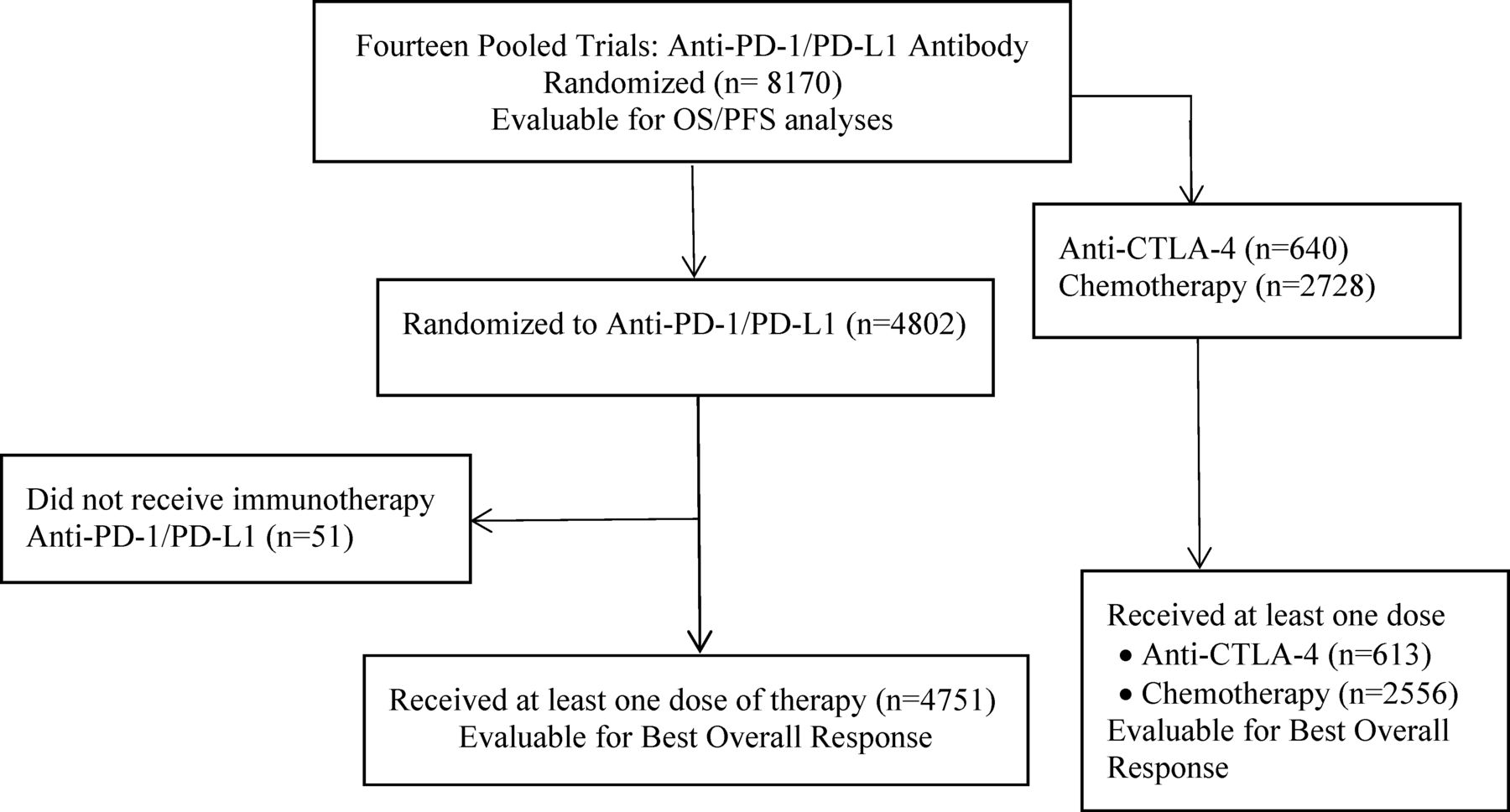
Comparison of iRECIST versus RECIST V.1.1 in patients treated with an anti-PD-1 or PD-L1 antibody: pooled FDA analysis | Journal for ImmunoTherapy of Cancer

iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics - The Lancet Oncology
Comparison of Radiological Tumor Response Based on iRECIST and RECIST 1.1 in Metastatic Clear-Cell Renal Cell Carcinoma Patient