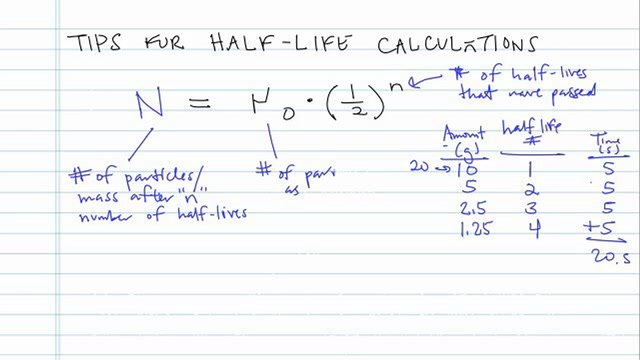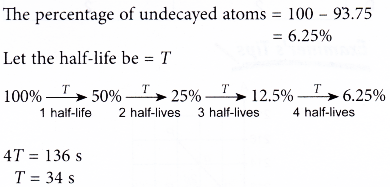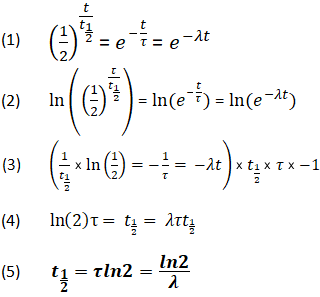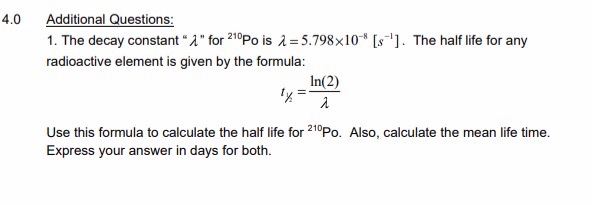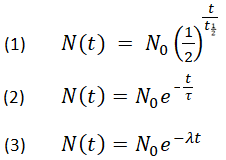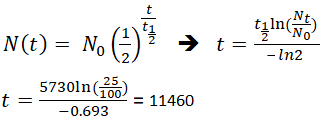Scientists measure half-life of element that's longer than the age of the universe | University of Chicago News
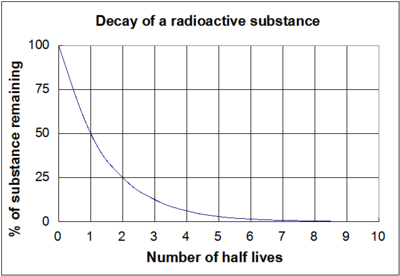
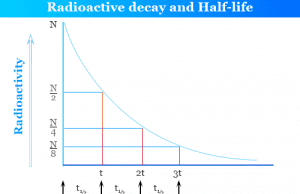
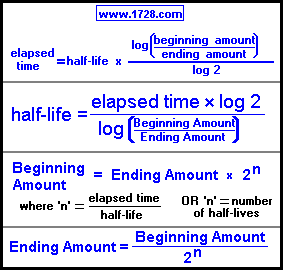
97. Half life of a radioactive element is 10 days ,the time during which quantity remains 0.1 of initial mass will be

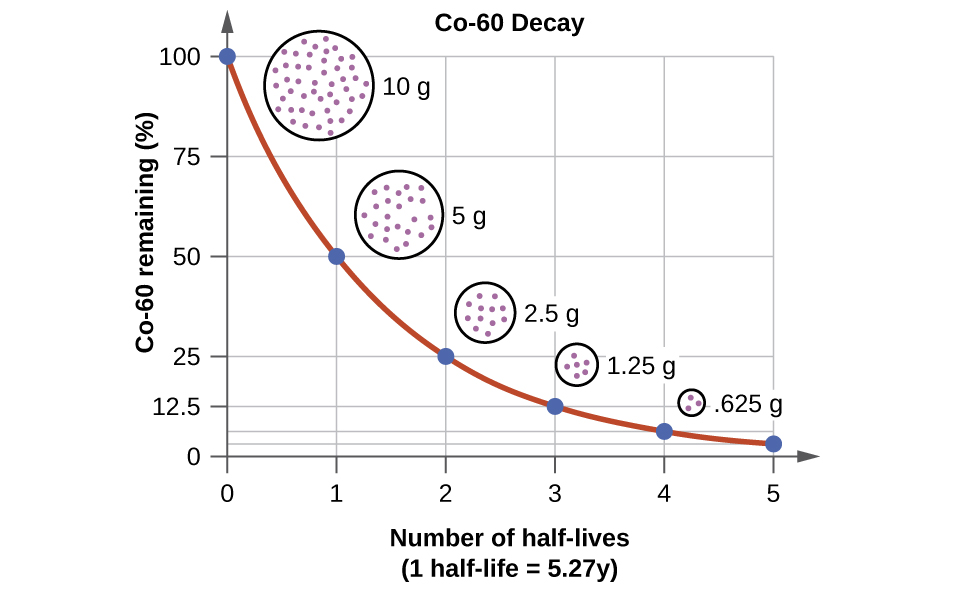
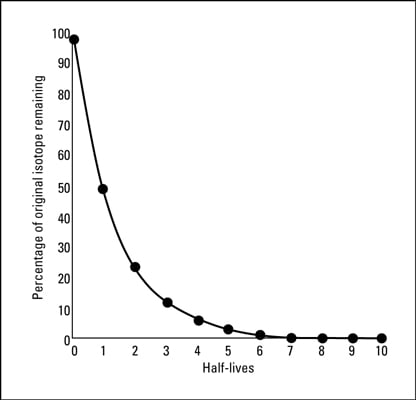
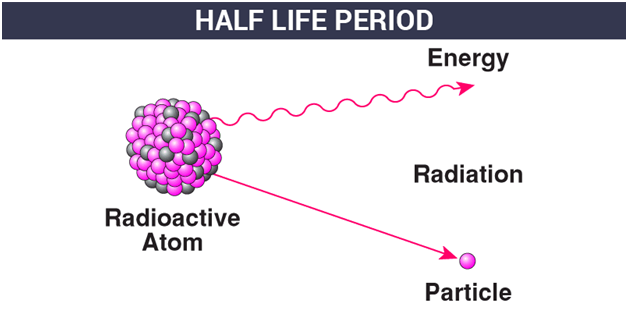
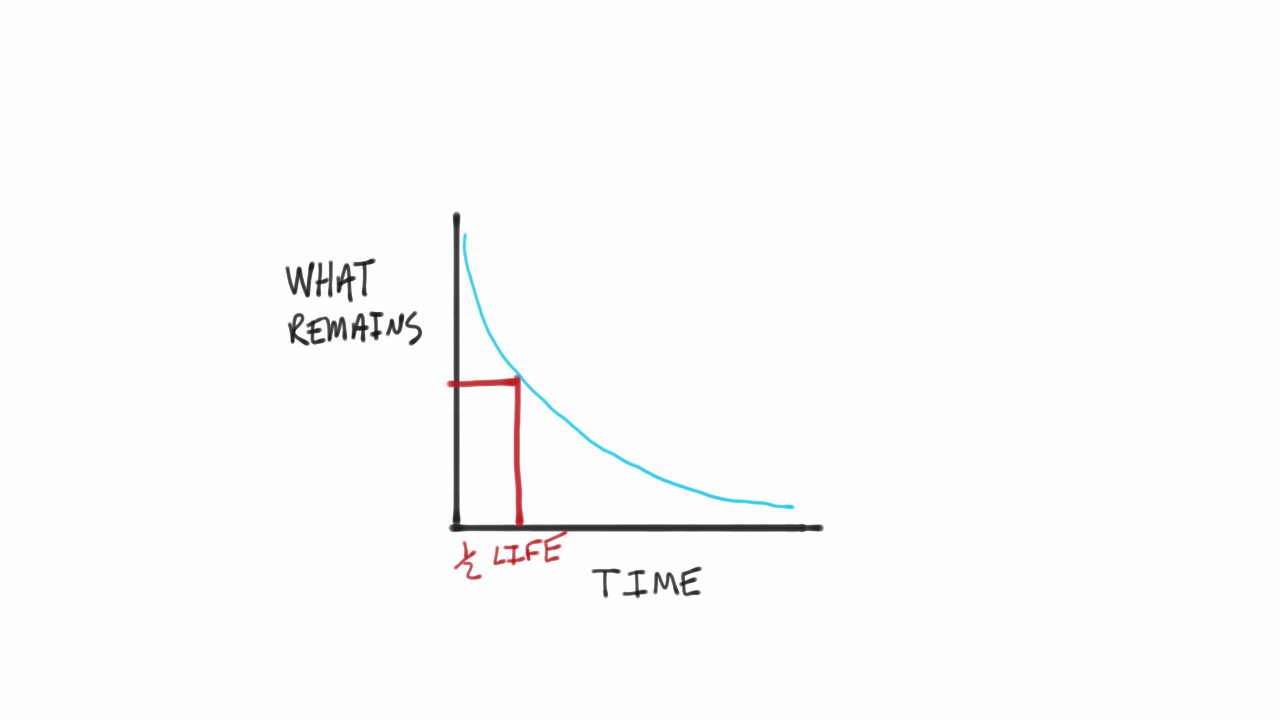
PPT - CALCULATING HALF-LIFE Aim: How can we measure how much of a radioactive element decayed? PowerPoint Presentation - ID:2129831

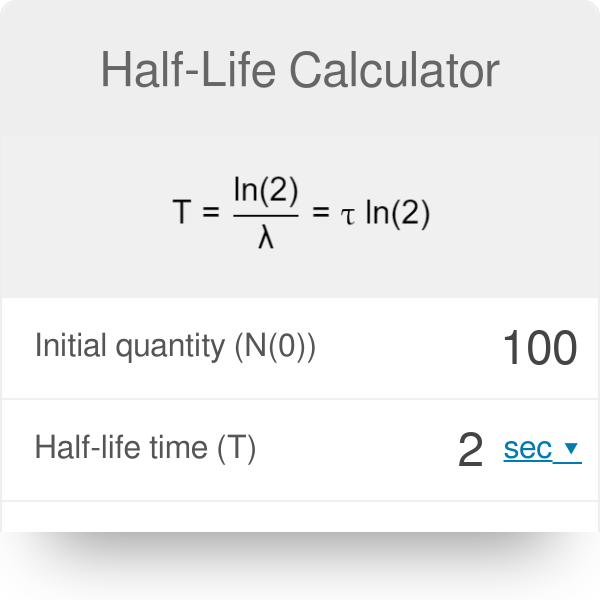
Half-life period of a radioactive element is 100 seconds. Calculate the disintegration constant and average life period. How much time will it take for 90% decay?