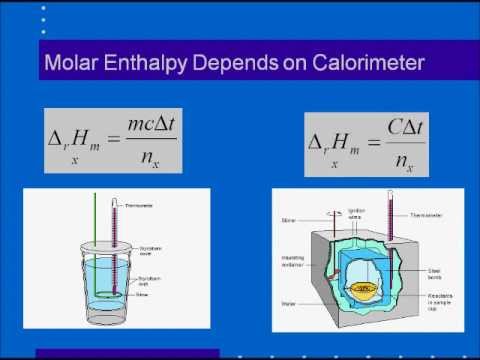
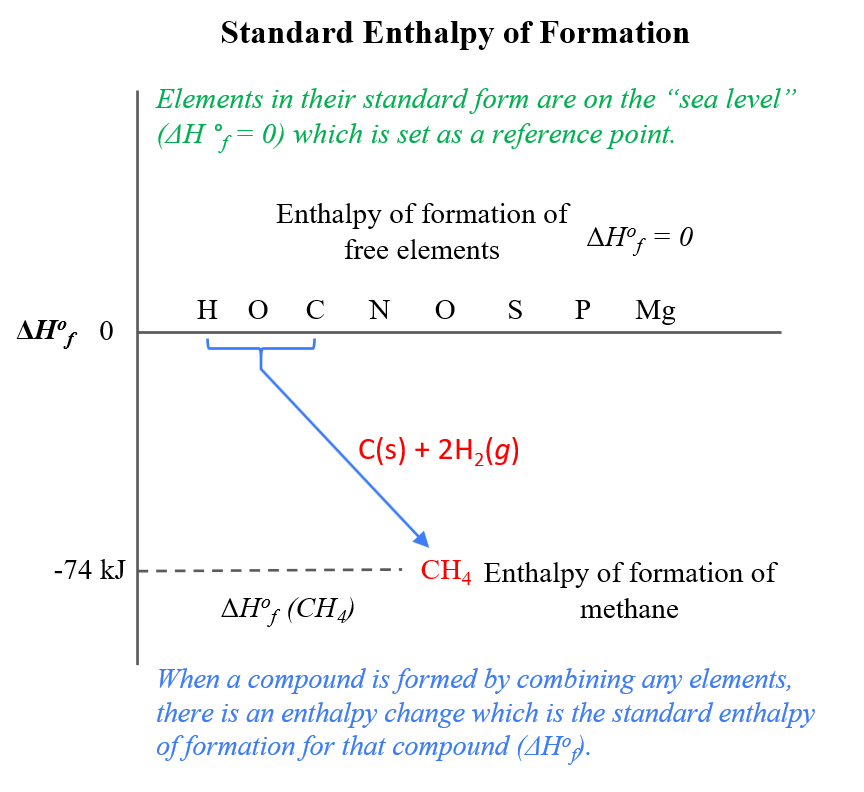
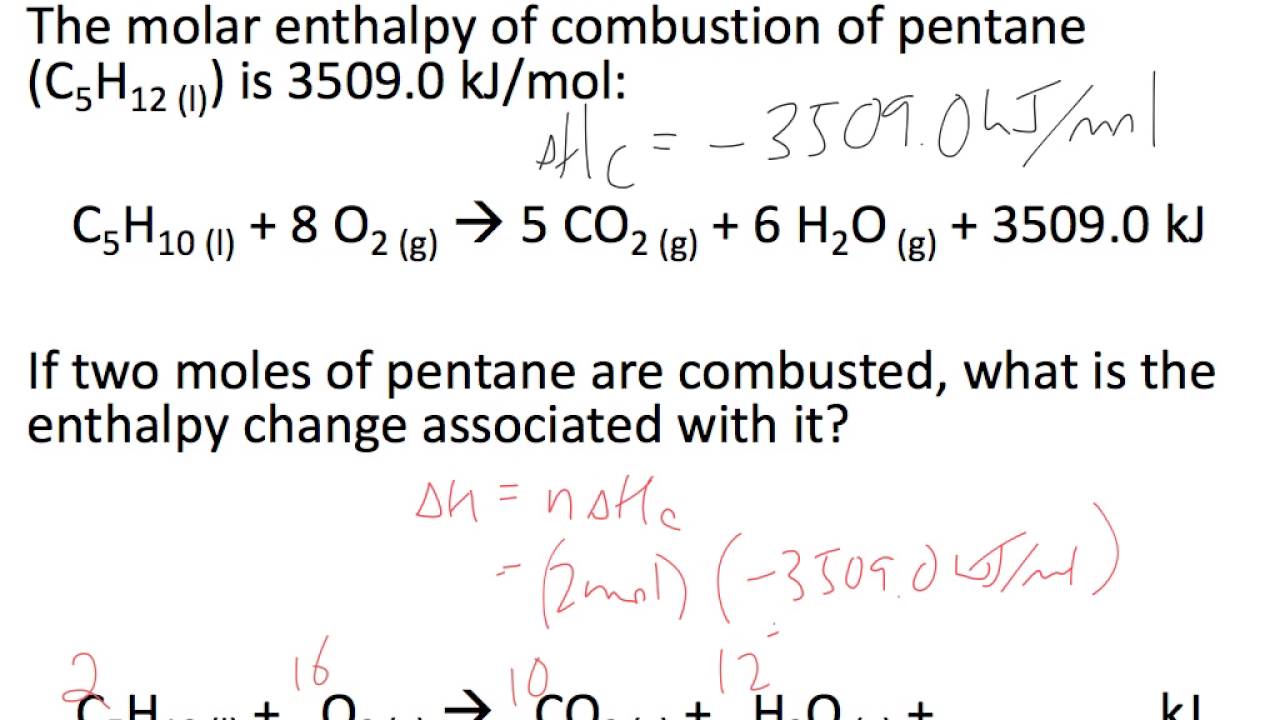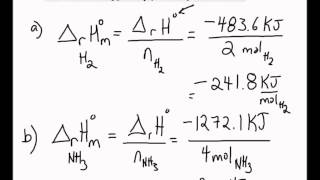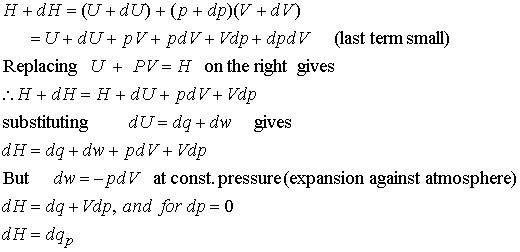Molar Enthalpies. use proper scientific terminology to describe molar enthalpies calculate molar enthalpies Calculate molar enthalpies using the. - ppt download

Calculate the lattice energy for the reaction: Li^+(g) + Cl^-(g)→ LiCl(s) Given: ΔHsub(Li) = 160 ; ΔHdiss(Cl2) = 244 ; IP(Li) = 520 ; EA(Cl) = - 365 and ΔHf(LiCl) = - 400 (all in kJ mole^-1 )

STANDARD MOLAR ENTHALPY OF FORMATION Enthalpy change when 1 mol of species is formed in its Standard State at a Specified Temperature from the most stable. - ppt download
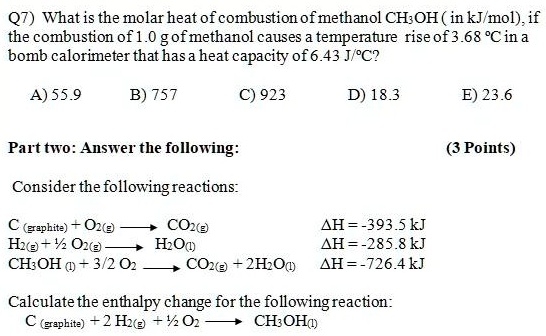
SOLVED: Q7) What is the molar heat of combustion of methanol CH;OH ( in kJmol). if the combustion of 1.0 gof methanol causes temperature rise of 3.68 %Cin a bomb calorimeter that

SOLVED: The molar enthalpy change for the neutralization reaction: HCI (aq) + NaOH (aq) +> NaCl (aq) + HzO (I) is AH=-57.3 k/mol. This is an exothermic reaction which means heat is

3:04 calculate the molar enthalpy change (ΔH) from the heat energy change, Q - TutorMyself Chemistry

Calculate the change in molar entropy and change in Gibbs' free energy when 1 mol of liquid butanol vaporizes at 25ºC to a gas that is at 0.001 atm? | Socratic