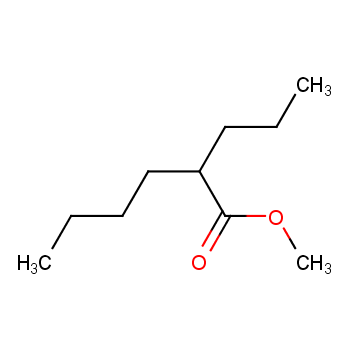
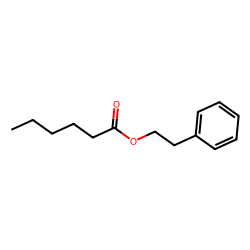
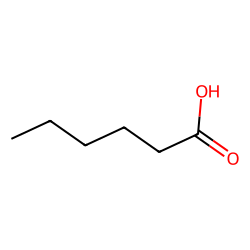
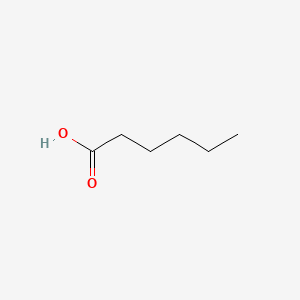
SOLVED: 14.8 Identify the compound in each group that is most soluble in water: Explain. a. butanone, butanoic acid, butane b. ethanoic acid (acetic acid), hexanoic acid, octanoic acid 14.9 Write the
Hexanoic acid,1,1'-[2,2-bis[[(1-oxohexyl)oxy]methyl]-1,3-propanediyl] ester supplier | CasNO.7445-47-8

a) CH_3 CH_2 CH_2 CH_3 \ or \ CH_3 CH_2 Co_2 H \\ (B) CH_3 (CH_2)_4 COOH \ or \ CH_3 (CH_2)_4 COO^- Na^+ 1. Which compound in each pair is more