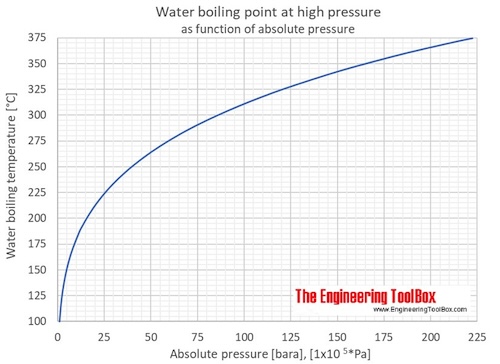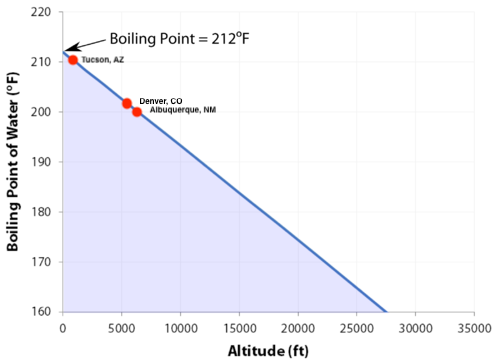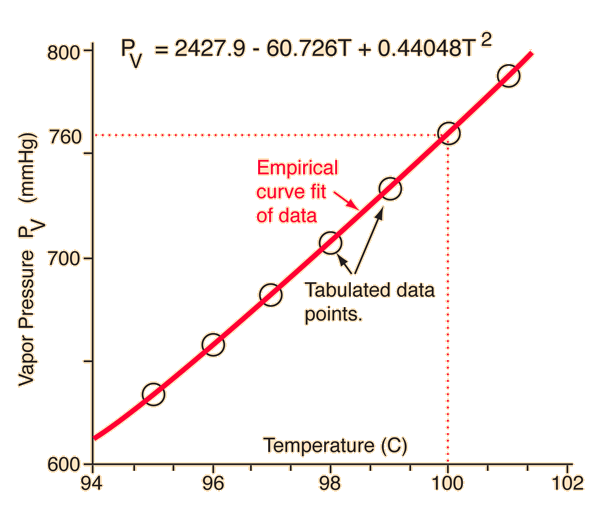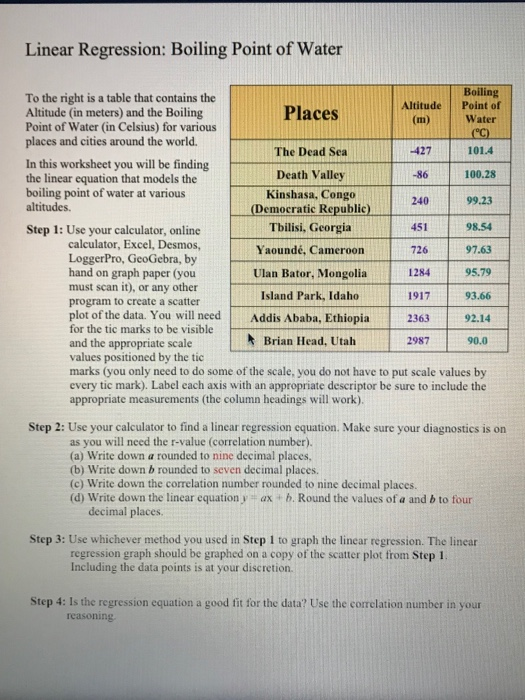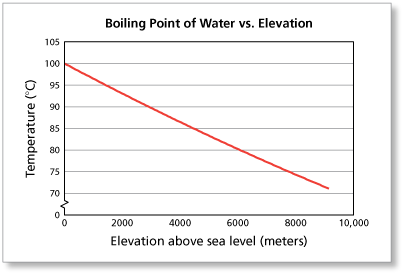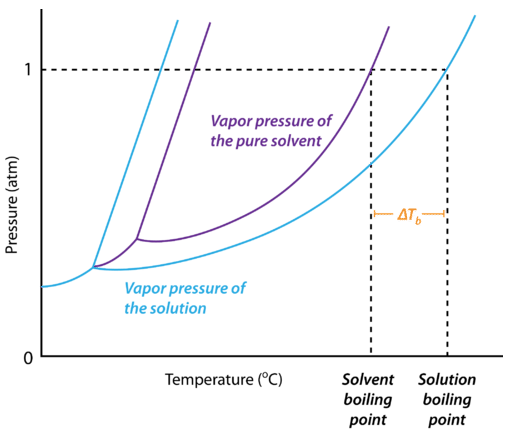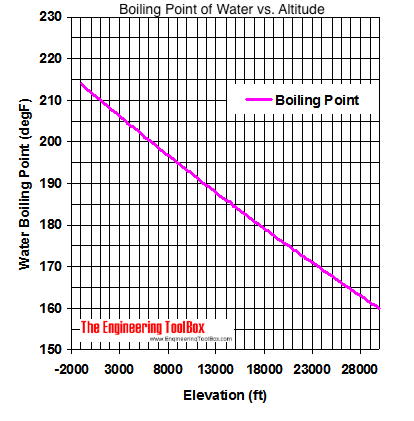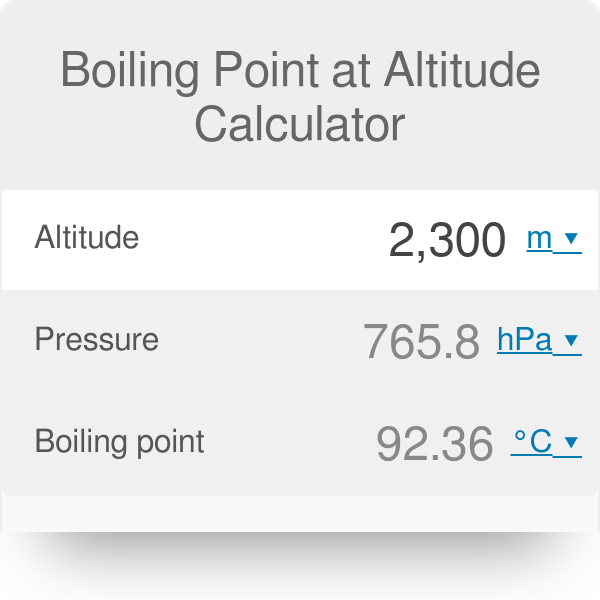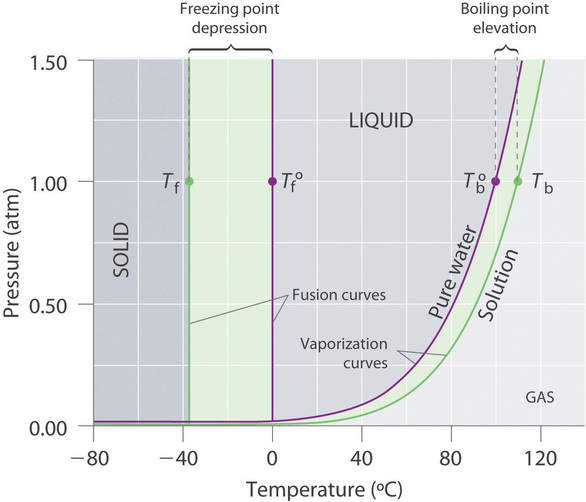
13.8: Freezing-Point Depression and Boiling-Point Elevation of Nonelectrolyte Solutions - Chemistry LibreTexts
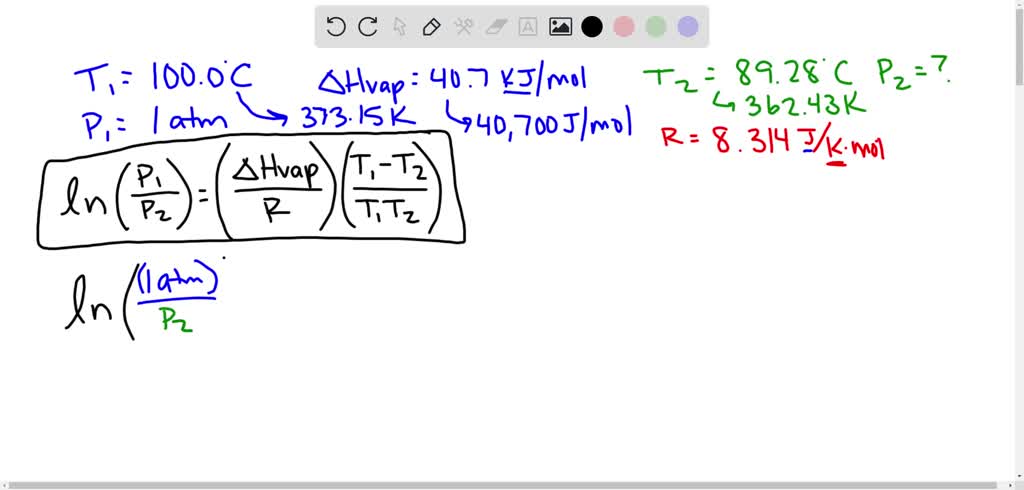
SOLVED: At sea level, the boiling point of water is 100'C. Estimate the vapor pressure of water at 110*C, The Heat of vaporization for water iS 40.7 kJmol-1 Use Clausius-Clapeyron equation and