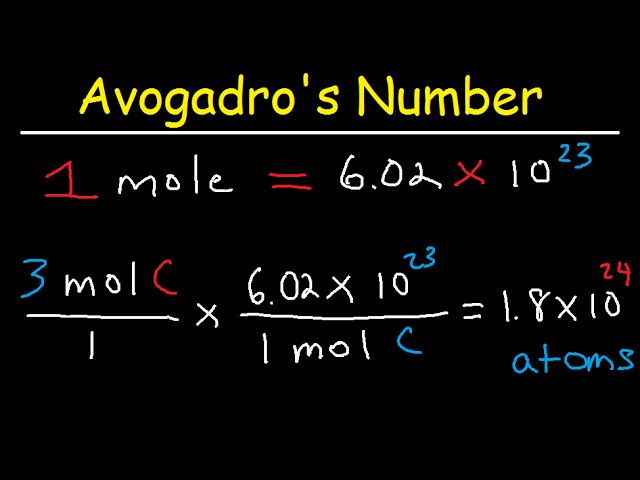Use Avogadro's Number to Calculate the Mass of a Single Atom | Relative atomic mass, Chemistry, Atom

Section 2 - Avogadro's Number and the Mole - Part 1 | Math Tutor DVD - Online Math Help, Math Homework Help, Math Problems, Math Practice!

Physical Chemistry #1: Relative Mass, the Mole and Avogadro's Constant (Slides & Student Led Tasks) | Teaching Resources

1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5

1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5
Clip of M1.3: The Mole Part 1 (Avogadro's Number) - SchoolTube - Safe video sharing and management for K12

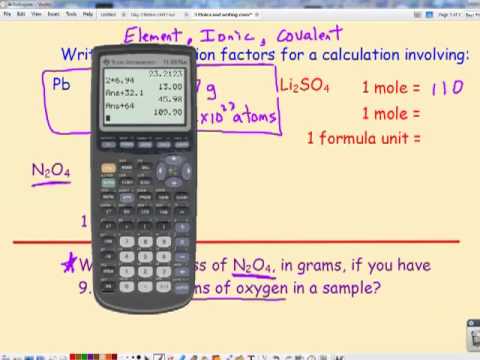
1 CALCULATOR USE FOR SCIENTIFIC NOTATION PUT INTO SCIENTIFIC NOTATION MODE TO GET ANSWERS IN SCI NOT PUT INTO NORMAL MODE TO GET DECIMAL NUMBERS. - ppt download

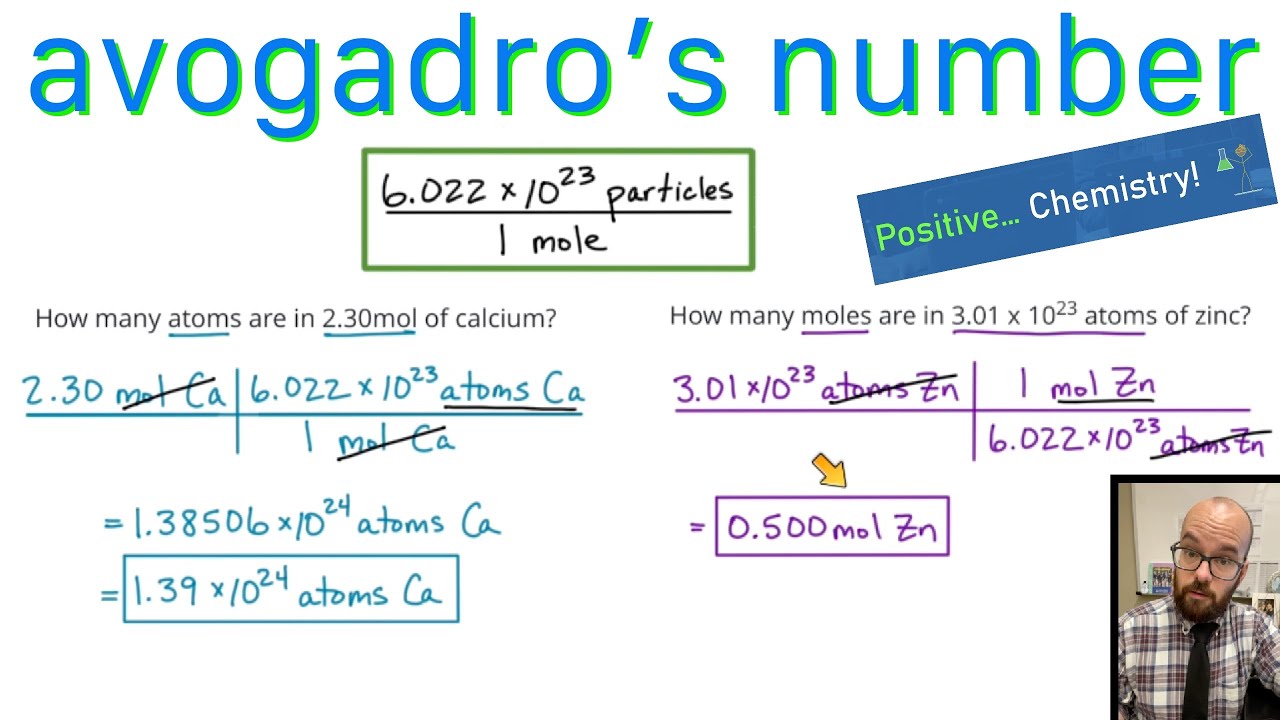
Objective: To reintroduce the mole and introduce two mole conversions Do Now: Solve each proportion = x2. 10 = x. - ppt download