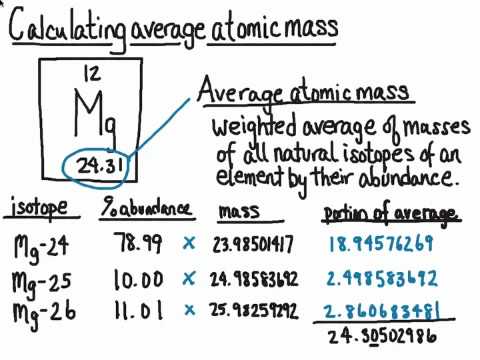
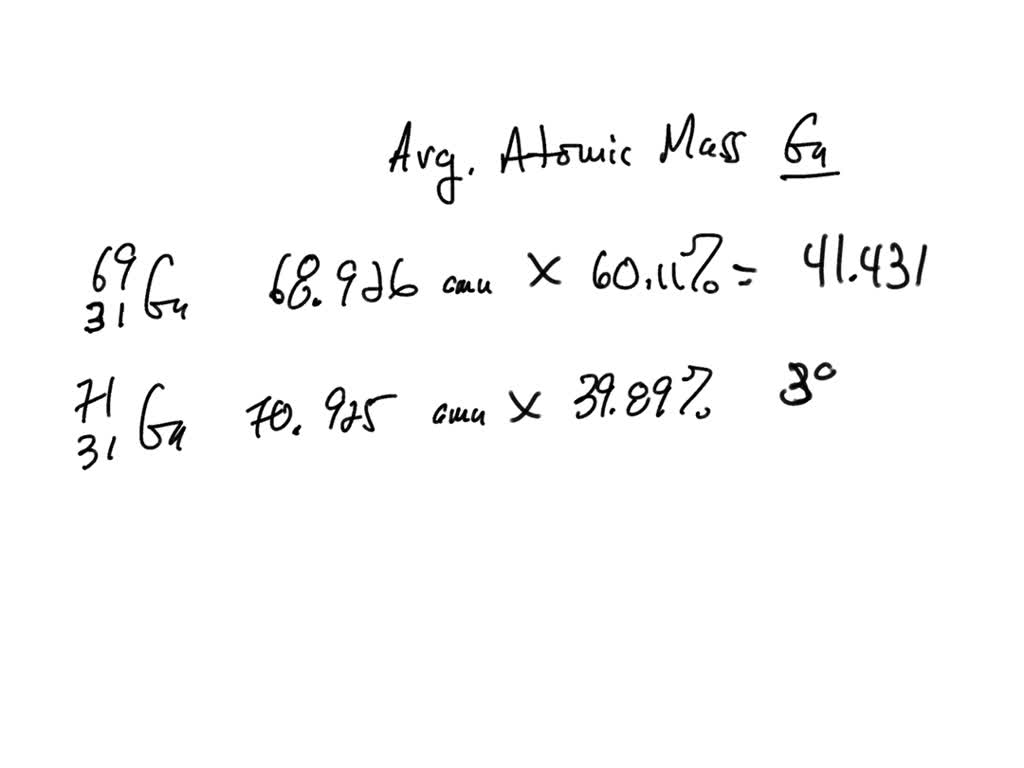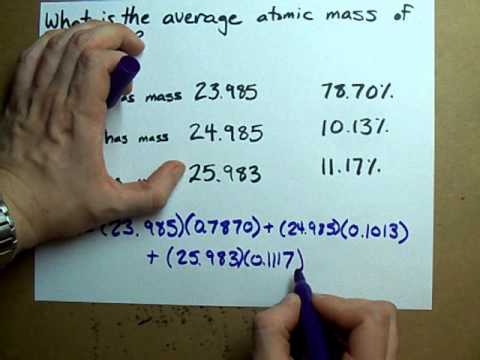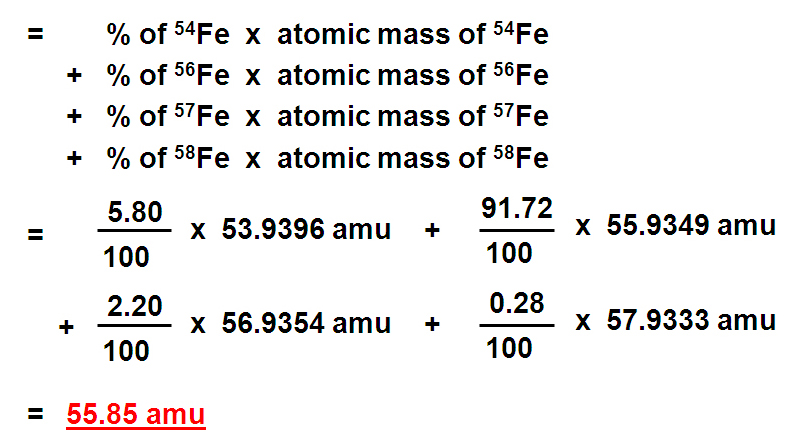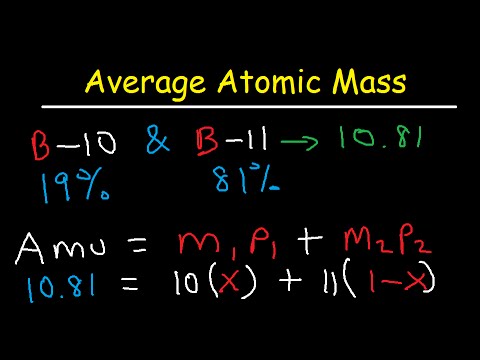Oxygen occurs in nature as a mixture of isotopes ^16O , ^17O and ^18O having atomic masses of 15.995 u, 16.999 u and 17.999 u and relative abundance of 99.763

How to Calculate Average Atomic Mass From Percent Abundance Examples, Practice Problems, Shortcut - YouTube