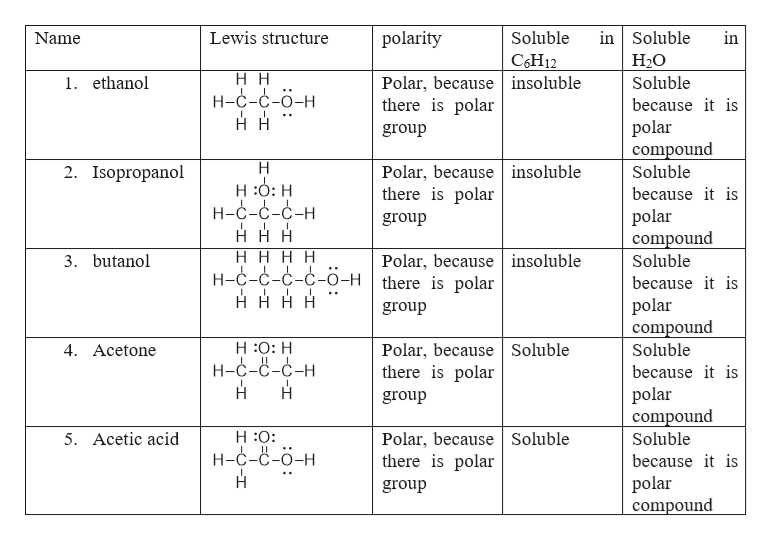
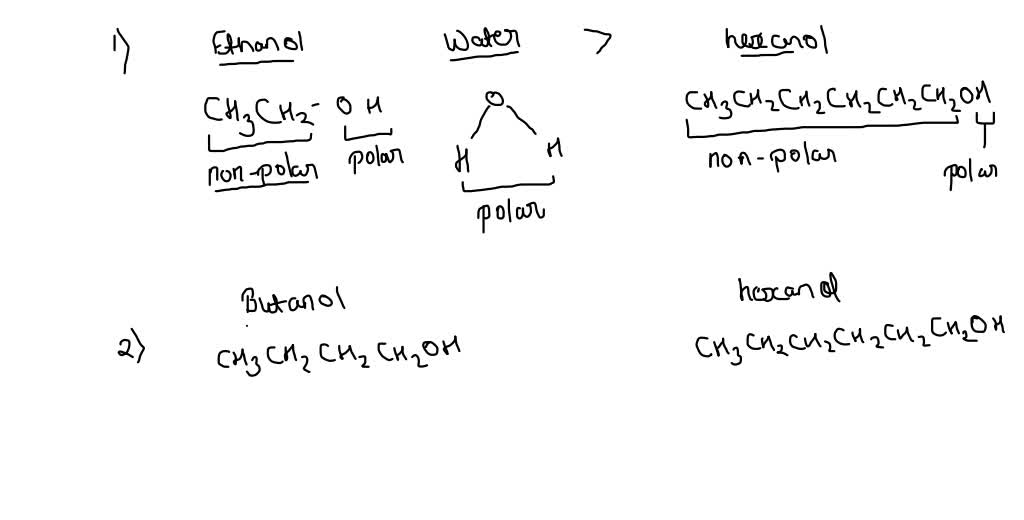
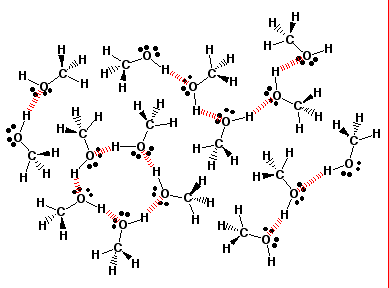
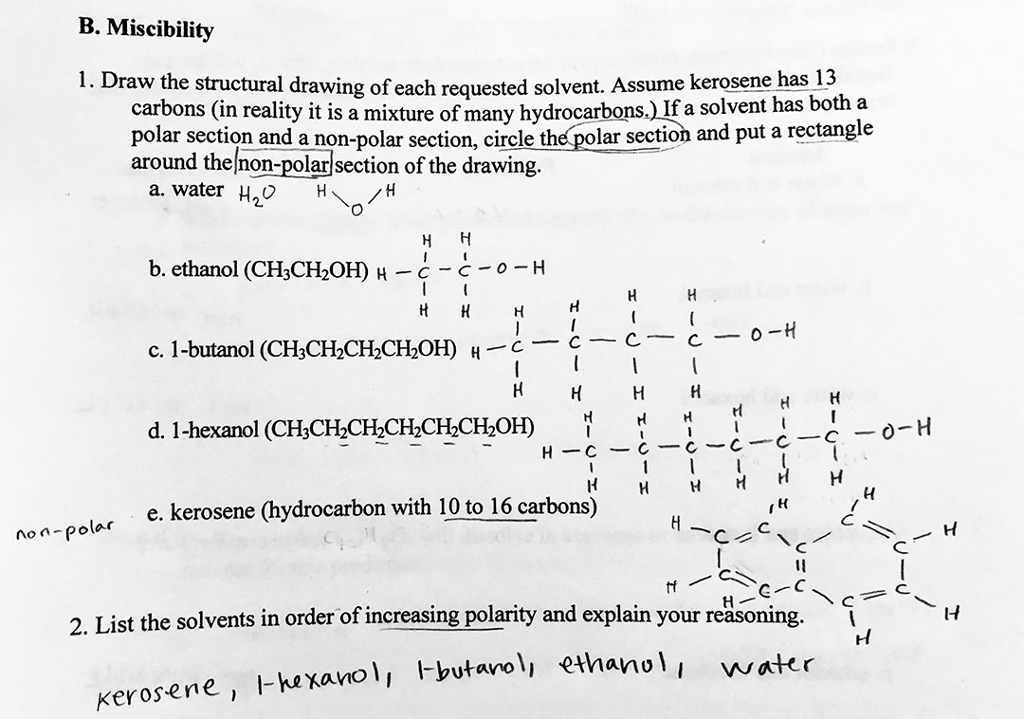
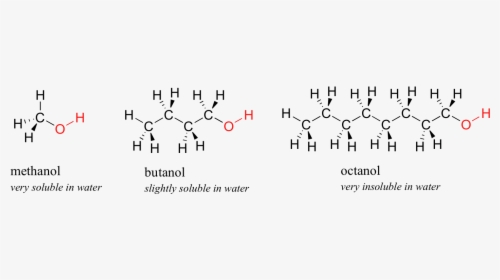
Is 1-butanol an ionic, molecular nonpolar, or molecular polar compound? What intermolecular forces are present? | Homework.Study.com

SOLVED: Ethanol is soluble in both water and toluene. Explain why this is unusual behavior: Use the concept of polarity in your answer: Explain why the solubility of I-pentanol in water at